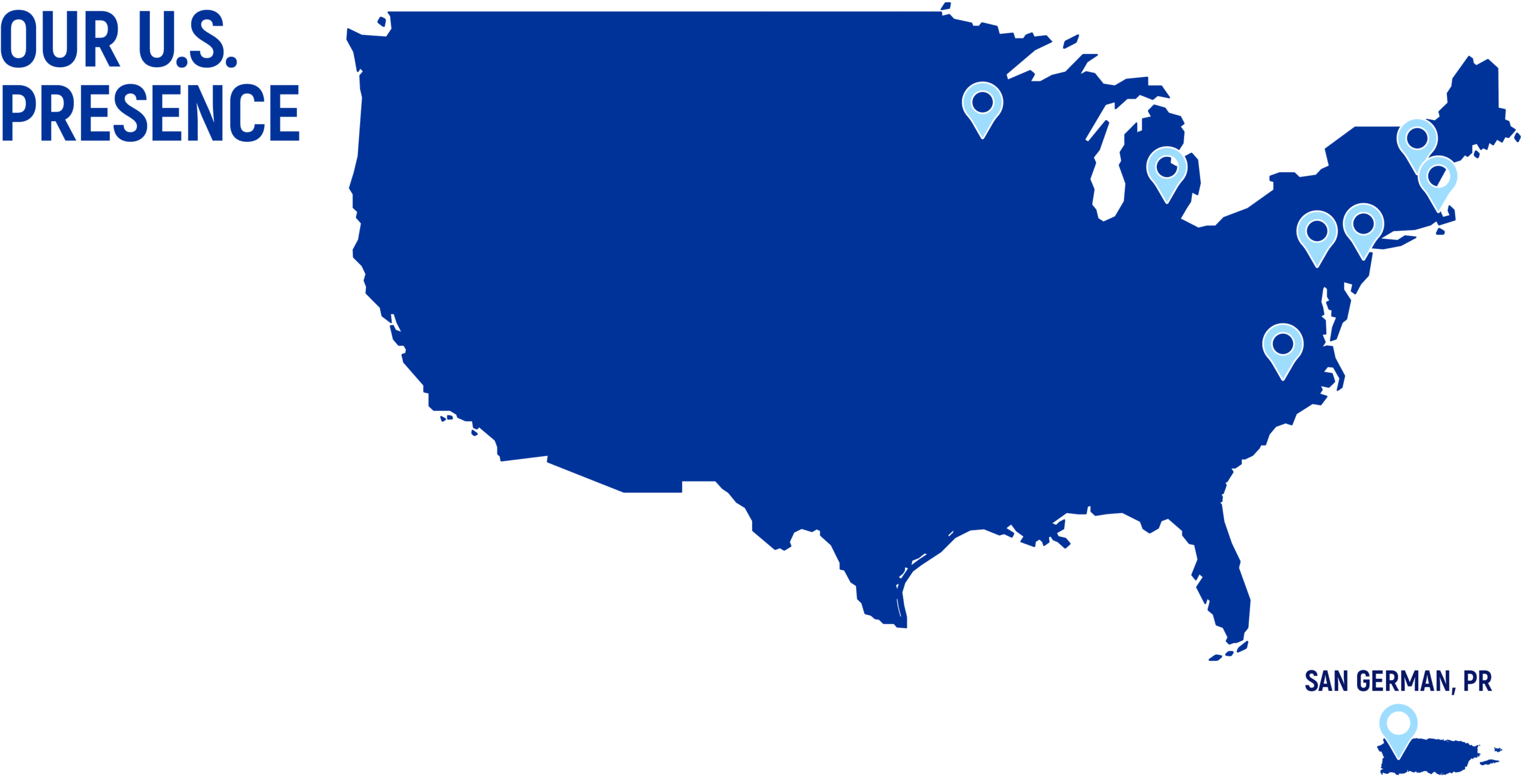
Boston
Advances small-molecule, biologics, and gene-therapy candidates from early pre-formulation through clinical phases. Previously known as Wolfe Labs.
Location: 19 Presidential Way, Woburn, MA 01801
Function: CDMO
Lab size: 22,000 ft²
Certifications / Registrations: GLP compliant
Services:
- Pre-formulation characterization of novel molecules
- Formulation development
- Full analytical support
- Tox supplies
- Lyophilization process development

Ann Arbor
Supports liquid and topical drug products from formulation development through non-sterile clinical supply manufacturing. Previously known as Velesco pharma.
Location: 28036 Oakland Oaks Ct., Wixom, MI 48393
Function: CDMO
Lab size: 16,000 ft²
Certifications / Registrations: GMP manufacturing facility
Services:
- Formulation development
- Analytical development
- Non-sterile clinical supply manufacturing
- Liquid-in-capsule filling

Oakdale
Provides FDA-registered contract research and manufacturing support for small molecules, biologics, and gene therapies. Founded as the original Pace® Life Sciences laboratory, then later expanded through the acquisition of P3 Scientific, Inc.
Location: 1311 Helmo Avenue North, Oakdale, MN 55128
Function: CRO
Lab size: 60,000 ft²
Certifications / Registrations: FDA-registered; DEA-registered
Services:
- Raw materials clearance programs
- Methods development and validation
- In-process and finished product release testing
- ICH stability programs
- Reference standards programs
- Extractables and leachables
- Full microbiology laboratory support (sterile and non-sterile product support)

Research Triangle Park
Provides FDA-registered CRO laboratory support for life sciences clients, from the Research Triangle Park area. Previously operated by Catalent.
Location: 160 N Pharma Drive, Morrisville, NC 27560
Function: CRO
Lab size: 63,000 ft2
Certifications / Registrations: GMP-aligned laboratory practices, FDA-registered; DEA-registered
Services:
- CRO laboratory services
- Life sciences laboratory support

Salem
Sterile Fill-finish Center of Excellence. Delivers clinical manufacturing, including sterile fill-finish, and clinical packaging and kitting, from a GMP facility in NH. Previously known as Bio-Concept Laboratories.
Location: 13 Industrial Way, Salem, NH 03079
Function: CDMO
Lab size: 30,000 ft²
Certifications / Registrations: GMP-compliant; aseptic processing capability
Services:
- Formulation development
- Process development
- Full analytical support
- Aseptic manufacturing for ophthalmic, otic and injectable products
- Toxicology-enabling materials
- Phase 1 and Phase 2 clinical trial materials
- Niche commercial manufacturing

Lebanon
Supports small- and large-molecule analytical services, formulation development and early-stage drug product manufacturing. Previously known as Whitehouse Analytical Laboratories and operated by Curia.
Location: 291 US Highway 22, Lebanon, NJ 08833
Function: CRO
Lab size: 40,000 ft²
Certifications / Registrations: GMP analytical laboratory, FDA-registered; DEA-registered
Services:
- Small- and large-molecule analytical services
- Formulation development
- Early-stage drug product manufacturing
- Analytical chemistry
- Microbiology
- Container closure integrity testing (CCIT)
- Sterility
- Packaging and delivery testing

Philadelphia
Delivers small-molecule formulation, preclinical and clinical manufacturing from a GMP clinical facility. Previously known as Emerson Resources, Inc.
Location: 600 Markley St., Norristown, PA 19401
Function: CDMO
Lab size: 33,000 ft²
Certifications / Registrations: GMP-certified; licensed for controlled substances
Services:
- Small-molecule formulation development and preclinical support
- Clinical manufacturing (tox supplies and Phase I/II clinical supplies)
- Tablets, capsules, liquids, suspensions, topical creams, gels and ointments
- Full analytical support
- Controlled substances and potent compounds
- Solvent processing
- Solubility enhancement (spray-drying, nano-suspensions, hot-melt extrusion)
- Drug product release and ICH stability studies
San Germán
Provides FDA- and DEA-registered analytical laboratory and manufacturing support services for pharmaceutical and medical device clients in Puerto Rico.
Location: San German, San German, PR 966
Function: CRO
Lab size: 22,000 ft²
Certifications / Registrations: FDA-registered; DEA-registered
Services:
- Chemistry and microbiology laboratories
- Raw materials clearance programs
- In-process and finished product testing
- ICH stability programs
- Facility and environmental monitoring programs
- Cleaning verification and disinfectant efficacy studies

Get in touch
Whether you’re shaping early plans or preparing to scale, our teams are here to help you move forward with clarity and confidence. Start the conversation and we’ll get back to you quickly.
